广西作为全国铝工业核心产区,2024年氧化铝产量达1454.30万吨,年产赤泥(Red Mud,RM)副产物逾1500万吨。赤泥因强碱性(pH=10-13)及高盐特性引发土壤板结、地下水污染等生态风险[1-2],其规模化堆存已成为制约行业绿色低碳发展的关键瓶颈。现行赤泥利用技术(选铁、建材掺合、有价元素提取等)普遍存在技术路线单一、处理成本高昂、产品附加值低等问题[3],在"双碳"战略背景下亟需开发经济高效的赤泥减量化与资源化技术体系。
赤泥资源化的核心难点在于碱脱除与多组分协同利用:1)传统脱碱技术难以兼顾处理效率与经济性,导致大量赤泥仍以低端建材形式消耗。例如,将赤泥与粉煤灰等材料结合,可将铝铁元素转化为复合絮凝剂,剩余残渣转化为复合白炭黑产品[4]。2)现有有价元素提取工艺(炼铁-炉渣浸出法、碱还原焙烧-酸浸法等)对复杂成分适应性差,存在金属回收率低、流程冗长、二次污染等问题[5]。因此,赤泥的资源化利用受制于工艺成本与产品性能,尚未形成规模化高值利用路径,亟需开发兼具高效脱碱与多组分协同增值的低成本工艺。
类水滑石(LDH)是一种阴离子插层层状材料,其化学组成通式可表示为M2+1-xM3+x(OH)2(An-)x/n‧mH2O,其主体层板由二价金属离子(Mg2+、Ca2+、Cu2+、Ni2+等)和三价金属离子(Al3+、Fe3+等)与氧原子形成的八面体,通过共边连接形成层间有可交换的阴离子An-(CO32-、NO3-、SO42-等阴离子)[6]。LDH在诸多领域具有广泛的应用,如CO2捕集[7]、磷回收[8]和固体碱多相催化[9]。作为一种具有二维层状结构的材料,因其可调层板组成与层板/间限域效应,为赤泥多组分的高值化利用提供了理想载体,在赤泥基LDH的合成中表现出了优异的潜力。研究表明[10],赤泥中Fe、Ca、Al、Ti等元素与LDH层板组分高度匹配,可作为合成LDH的前体,通过不同合成方法获得赤泥基LDH材料。例如,利用赤泥与MgO或MgCl2·6H2O合成的MgAlFe-LDH材料可作为高分子聚合物的阻燃剂[11]。利用硼泥、电石渣或镍铁炉渣等工业固废代替传统化学试剂,与赤泥协同作用,能够合成赤泥基Fe/Al-LDH[12]、Ca/Al-LDH[13]、Mg/Al-LDH[14]或Mg/(Al-Fe)-LDH[15]材料,在磷酸盐或CO2吸附等领域取得良好效果。此外,赤泥附着的NaOH提供原位碱源调节LDH合成过程中的pH值,无须添加反应助剂。然而,现有制备工艺多依赖外源金属试剂,需酸浸/煅烧等预处理步骤,工序繁琐且能耗高,导致成本堆高。因此,探索一种不需外加金属源、低成本、操作简单且易于工业化生产的赤泥基LDH制备方法至关重要。
目前,合成LDH的主流方法包括液相合成法(如共沉淀法[16]、水热法[17]、溶胶-凝胶法[18]等)和固相合成法(如固相煅烧法[19]、机械化学法[20])。液相合成法通常需要控制反应体系的pH值,且需要消耗大量水和盐类,可能引起二次污染。因此,迫切需要开发一种简单、经济且环保的LDH制备方法,尤其是以低成本原料为基础的技术。
机械力化学合成法(Mechanochemical synthesis,MCS)是一种以高能球磨为核心的固相反应技术,被认为是制备各类复合材料的一种高效绿色方法[21]。与传统液相法(高耗水、二次污染)与固相煅烧法(高能耗)相比,MCS具备工艺简单、能耗低、污染小、成本低以及适于规模化生产等优势。已有研究表明,通过MCS可实现无外源试剂的LDHs绿色合成,具有显著的经济和绿色环保优势。例如,Stepanova[22]等人通过机械能驱动矿物相重构合成MgAl-LDH,验证了该方法的可行性。
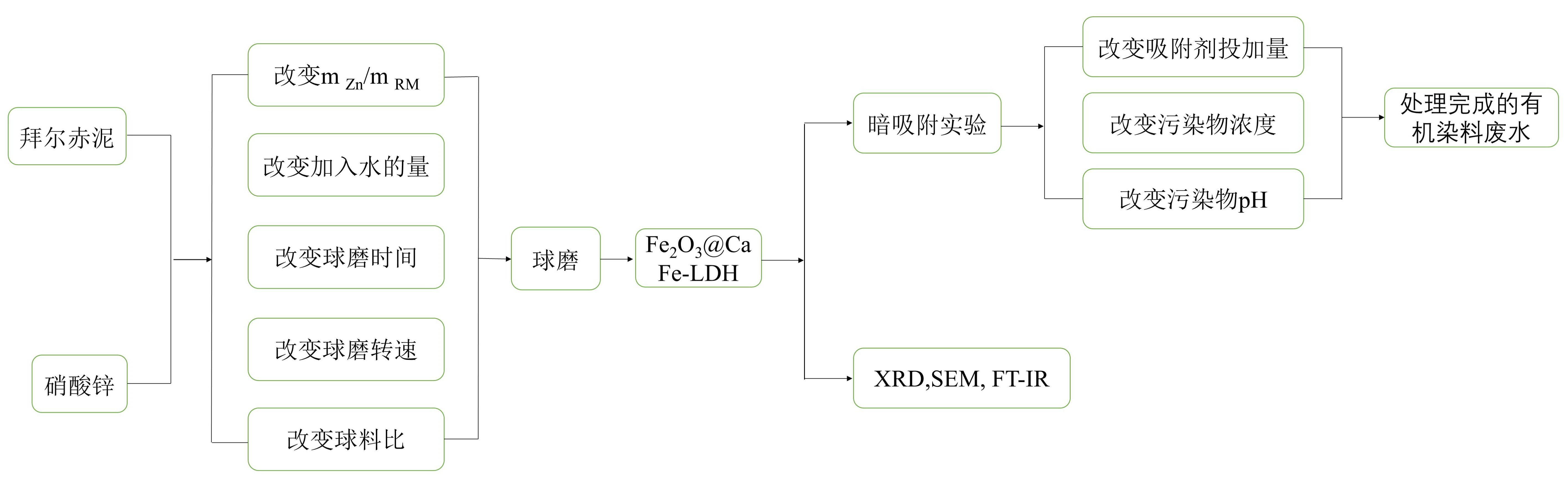
本作品突破传统“酸浸-外源补金属”合成路线,基于赤泥组分与LDH晶格的高度匹配性,创新性提出机械化学法原位构筑Fe2O3@CaFe-LDH异质结复合材料。通过工艺参数(球磨能量密度、固液比、作用时间)优化,阐明矿物相重构动力学与LDHs晶体生长规律的关联性,形成低成本、低能耗、低排放的工业化可行技术原型。系统研究表明,Fe2O3@CaFe-LDH的最佳制备条件为:赤泥与Zn(NO3)2·6H2O的质量比(m RM:m Zn)为1:2,湿磨时间4 h,水用量2 mL,球粉质量比50:1,球磨速度250 rpm。以Fe2O3@CaFe-LDH异质结复合材料作为低成本高效的有机染料吸附剂,研究其对有机染料靛蓝的吸附性能。实验结果表明,Fe2O3@CaFe-LDH对50 mg/L靛蓝溶液在120 min内实现100%脱色。本作品为赤泥的高值化利用及染料废水的绿色处理提供了一种全新的解决方案,兼具环境保护和资源化利用的双重意义,对于推动冶金工业的绿色可持续发展具有重要的应用价值和广阔的应用前景。
参考文献:
[1]Li R B, Zhang T G, Liu Y, et al. Calcification–carbonation method for red mud processing [J]. Journal of Hazardous Materials, 2016, 316: 94-101.
[2]Mian M. Ahson Aslam, Sun T T, Dai M, et al. Technologies for recovery of iron from red mud: Processes, challenges and opportunities [J]. Sustainable Materials and Technologies, 2024, 41: e01053.
[3]Ma S L, Liu X M, Zhang Z Q, et al. Removal, conversion and utilization technologies of alkali components in bayer red mud [J]. Journal of Environmental Management, 2025, 373: 123781.
[4]Li Z F, Zhang J, Li S C, et al. Feasibility of preparing red mud-based cementitious materials: Synergistic utilization of industrial solid waste, waste heat, and tail gas [J]. Journal of Cleaner Production, 2021, 288:124896.
[5]Pan X L, Wu H F, Lv Z Y, et al. Recovery of valuable metals from red mud: A comprehensive review [J]. Science of The Total Environment, 2023, 904: 166686.
[6]Tang S, Lee H K. Application of dissolvable layered double hydroxides as sorbent in dispersive solid-phase extraction and extraction by co-precipitation for the determination of aromatic acid anions [J]. Analytical Chemistry, 2013, 85(15): 7426-7433.
[7]Shang S, Hanif A, Sun M, et al. Novel M (Mg/ Ni/ Cu)-Al-CO3 layered double hydroxides synthesized by aqueous miscible organic solvent treatment (AMOST) method for CO2 capture [J]. Journal of Hazardous Materials, 2019, 373: 285-293.
[8]Yang F, Zhang S, Sun Y, et al. Assembling biochar with various layered double hydroxides for enhancement of phosphorus recovery [J]. Journal of Hazardous Materials, 2018, 365: 665-673.
[9]Yu I K M, Hanif A, Tsang D C W, et al. Tuneable functionalities in layered double hydroxide catalysts for thermochemical conversion of biomass-derived glucose to fructose [J]. Chemical Engineering Journal, 2020, 383: 122914.
[10]Wang B T, Qu J, Li X W, et al. Precursor preparation to promote the adsorption of Mg-Al layered double hydroxide [J]. Journal of the American Ceramic Society, 2016, 99(9): 2845-3167.
[11]Qian Y, Li S S, Chen X L. Synthesis and characterization of LDHs using Bayer red mud and its flame-retardant properties in EVA/LDHs composites [J]. Journal of Material Cycles and Waste Management, 2015, 17: 646-654.
[12]He Y L, Wei G T, Wang N, et al. Novel S-scheme heterojunction of red mud-based Fe2O3/Co-Al-LDH for the photo-Fenton degradation of gatifloxacin under visible light [J]. Separation and Purification Technology, 2025, 369: 133160.
[13]Zhang P, Qian G R, Shi H S, et al. Mechanism of interaction of hydrocalumites (Ca/Al-LDH) with methyl orange and acidic scarlet GR [J]. Journal of Colloid and Interface Science, 2012, 365(1): 110-116.
[14]Hu P, Zhang Y H, Lv F Z, et al. Reduction in chemical oxygen demand of TNT red water using layered double hydroxide prepared from red mud and brucite [J]. Environmental Engineering Science, 2017, 34(10): 721-730.
[15]Yang Y Q, Gao N Y, Chu W H, et al. Adsorption of perchlorate from aqueous solution by the calcination product of Mg/(Al-Fe) hydrotalcite-like compounds [J]. Journal of Hazardous Materials, 2012, 209-210: 318-325.
[16]Iris K.M. Yu, Aamir Hanif, Daniel C.W. Tsang. Tuneable functionalities in layered double hydroxide catalysts for thermochemical conversion of biomass-derived glucose to fructose [J]. Chemical Engineering Journal, 2020, 383: 122914.
[17]Kubota K, Pang Y, Miuraro A, et al. Redox reactions of small organic molecules using ball milling and piezoelectric materials [J]. Science, 2019, 366(6472): 1500-1504.
[18]Farzana Rahman, Yang X Y, Fiona Motswaiso, et al. Effective synthesis of ethylene urea from CO2 adsorbed cerium doped Mg-Al layered double hydroxide [J]. Journal of Cleaner Production, 2024, 434: 140191.
[19]I. Crespo, C. Barriga, V. Rives, et al. Intercalation of iron hexacyano complexes in Zn,Al-hydrotalcite [J]. Solid State Ionics, 1997, 101-103(2): 729-735.
[20]Abbas Fahami, F.S. Al-Hazmi, A.A. Al-Ghamdi, et al. Structural characterization of chlorine intercalated Mg-Al layered double hydroxides: A comparative study between mechanochemistry and hydrothermal methods [J]. Journal of Alloys and Compounds, 2016, 683: 100-107.
[21]Qu J, Sha L, Wu C, et al. Applications of mechanochemically prepared layered double hydroxides as adsorbents and catalysts: a mini-review [J]. Nanomaterials, 2019, 9(1): 80.
[22]L.N. Stepanova, E.O. Kobzar, N.N. Leont’eva, et al. Study of the chemical and phase transformations in the mechanochemical synthesis of the MgAl-layered double hydroxide [J]. Journal of Alloys and Compounds, 2022, 890: 161902.